|
In his classic experiment, Thomson measured the mass-to-charge ratio of the cathode rays by measuring how much they were deflected by a magnetic field and comparing this with the electric deflection. How did J J Thomson measure the mass of cathode rays? How did J J Thomson contribute to the discovery of the electron?įurther research by Thomson, as well as work by Henri Becquerel (1852-1908), Lenard, Ernst Rutherford (1871-1937), and others, helped to show that the electron identified by Thomson was the same as the negatively charged particles observed in phenomena such as radioactivity and the photoelectric effect. In one experiment, Thomson attempted to use magnetism to see if negative charge could be segregated from cathode rays, in another he tried to deflect the rays with an electric field, and in a third he assessed the charge-to-mass ratio of the rays. What did Joseph John Thomson do with magnetism? 
Thomson realized that the accepted model of an atom did not account for negatively or positively charged particles. He demonstrated that cathode rays were negatively charged. Thomson discovered the electron by experimenting with a Crookes, or cathode ray, tube. This proved that the negative charge and the ray were inseparable and intertwined. This cylinder had two slits in it, leading to electrometers, which could measure small electric charges. Thomson’s First Cathode Ray Experiment His first experiment was to build a cathode ray tube with a metal cylinder on the end.  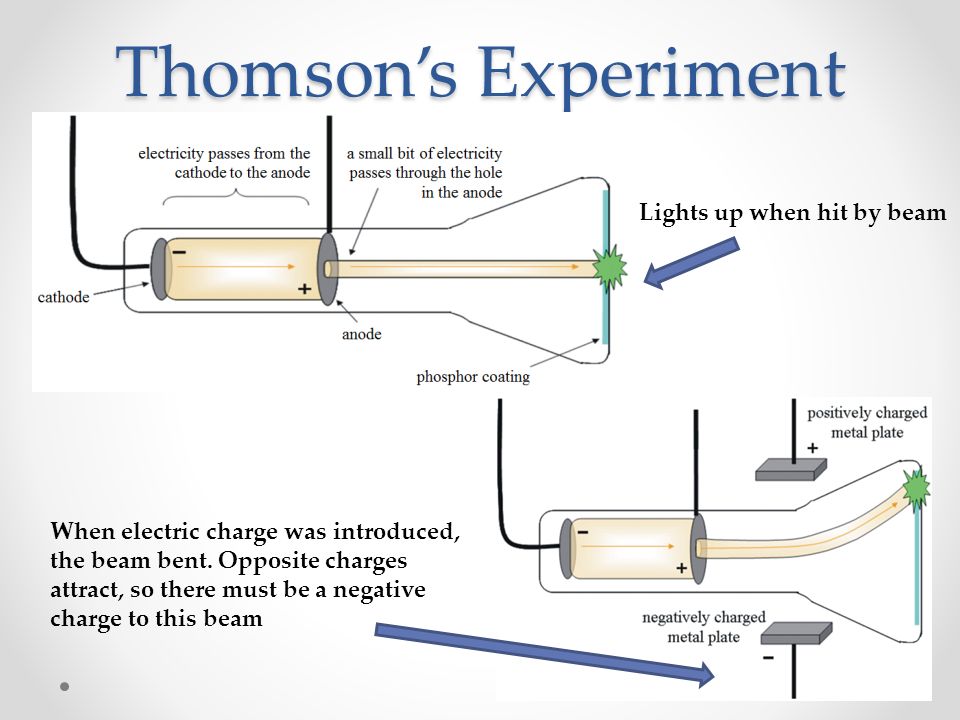
How did JJ Thomson perform his experiment?
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
